University of California, Los Angeles (UCLA) chemists are redesigning the atomic building blocks of organic solar cells to mimic nature – or more specifically, the way plants produce and store energy through photosynthesis.
According to the UCLA team, the materials used in current rooftop solar systems can only store energy from the sun for microseconds, but by tweaking the nanostructures of photovoltaic plastics into a more elegant, plant-like form, they found energy could be stored for several weeks.
“Biology does a very good job of creating energy from sunlight,” said Sarah Tolbert, a UCLA professor of chemistry and one of the senior authors of the research. “Plants do this through photosynthesis with extremely high efficiency.”
“In photosynthesis, plants that are exposed to sunlight use carefully organized nanoscale structures within their cells to rapidly separate charges — pulling electrons away from the positively charged molecule that is left behind, and keeping positive and negative charges separated,” Tolbert said. “That separation is the key to making the process so efficient.”
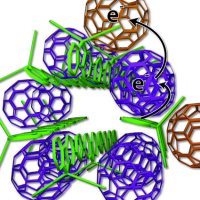
Photovoltaic plastic rapidly loses charge because its atomic structure resembles a “disorganised mass of polymer spaghetti”, which absorbs sunlight and passes electrons to “random fullerene meatballs”, creating an electric current. The disarray allows electrons to jump from the fullerene acceptor back to the polymer donor, and all current is lost.
By arranging the polymer spaghetti in bundles and precisely placing fullerene meatballs so they effectively toss electrons back and forth without touching the polymer, the UCLA system pulls the charges apart, ensuring a current that can last for weeks.
“When the charges never come back together, the system works far better,” said Benjamin Schwartz, a UCLA professor of chemistry and another senior co-author. “This is the first time this has been shown using modern synthetic organic photovoltaic materials.”
The technology is not only more efficient, it’s also environmentally-friendly. The nanostructures self-assemble simply by being in close proximity. This process can take place in water instead of the more toxic solutions currently used for producing organic solar cells.
“Once you make the materials, you can dump them into water and they assemble into the appropriate structure because of the way the materials are designed,” Schwartz said. “So there’s no additional work.”
The work, published in the journal Science, is part of a wider quest to find cheaper alternatives to silicon solar cell technology. Organic solar polymers hold great promise for low-cost solar power, being easier to produce and install, but efficiency levels are currently much lower than that of silicon PV cells.
Related : The Home Energy Storage Revolution