Researchers at the Massachusetts Institute of Technology (MIT), say they may have perfected a way to control the capture and release of solar energy through thermo-chemical technology, a method of harnessing solar power which was explored in the 1970s but abandoned as too expensive and impractical.
Conventional solar capture and storage technologies used today are primarily photovoltaic cells (solar panels), which convert sunlight directly into electricity; and solar thermal, which concentrate the sun’s heat and use it to boil water to turn a turbine, or use the heat directly for hot water or home heating.
Thermo-chemical solar power captures energy from sunlight and stores it as heat within molecules of a certain chemical. Then that energy, when needed, is released on demand. Unlike standard solar thermal systems which can allow heat to slowly leach away over time, heat stored within the thermo-chemical fuel remains stable.
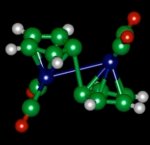
Unfortunately, the essential element needed to allow the thermo-chemical to both absorb sunlight and release it – ruthenium – was too rare to ever be cost effective. But the MIT research team say they have uncovered exactly how the essential molecule responsible for absorbing, storing, and releasing solar energy works. And this understanding, they said, should make it possible to find similar chemicals based on more abundant, less expensive materials than ruthenium.
According to Jeffrey Grossman, Associate Professor of Power Engineering in the Department of Materials Science and Engineering, the molecule – fulvalene diruthenium – absorbs solar energy, it goes into a higher-energy state where it can remain stable indefinitely. Then, triggered by a small addition of heat or a catalyst, it snaps back to its original shape, releasing heat in the process.
“It takes many of the advantages of solar-thermal energy, but stores the heat in the form of a fuel. It’s reversible, and it’s stable over a long term. You can use it where you want, on demand. You could put the fuel in the sun, charge it up, then use the heat, and place the same fuel back in the sun to recharge,” Professor Grossman said.
Our professional solar installers in Melbourne will assess and determine your energy needs. We customise a solar panel system in Melbourne to fit the roof size of your property, ensuring you receive the most suitable solar panel system for your Melbourne home, meeting to property’s energy requirements.